How Gerd Ulrich Nienhaus and his team make biological processes visible at the molecular level.
Research into molecular interactions within living cells, tissues and organisms forms the basis of countless developments in modern biomedicine. To develop new drugs, improved diagnostic methods and more efficient therapeutic approaches, the pharmaceutical industry and medical research need insights into cellular processes at the smallest level. "Modern light microscopy methods play a key role in this," says Prof. Gerd Ulrich Nienhaus, who has been tracking down biomolecules at the Institute of Applied Physics at KIT since 2009.
In the beginning, there is only one cell – it contains all the genetic information necessary for the development of a fully grown living being. In between, the cell divides itself many times. Countless processes must occur in precise coordination to ensure the development of an organism and the function of all its cells. "We are developing new light microscopy techniques and methods of specific fluorescence labeling that allow us to observe these fascinating molecular processes – and at a spatial resolution that was long thought to be unfeasible in principle," Nienhaus explains.
"I see my role as providing impetus for biomedical innovation. Research that seems rather abstract today may save countless lives in a few years."
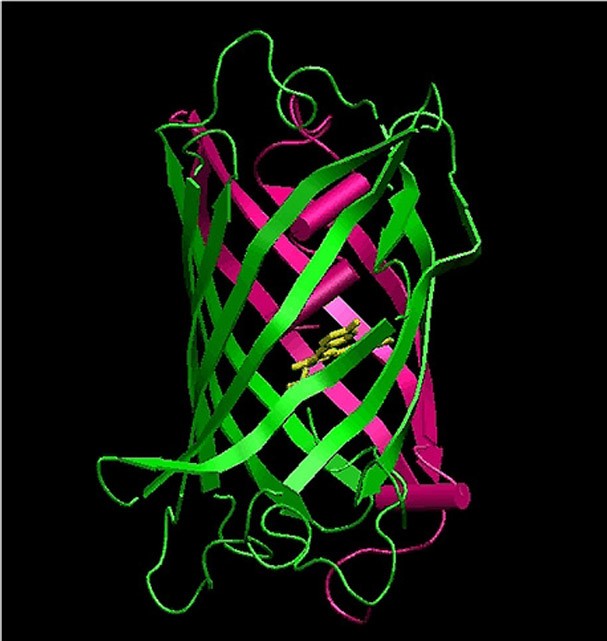
Essential to these studies is the ability to tag biomolecules and cellular structures with fluorescent markers that are specifically stimulated to glow by light irradiation. "The development of new marker technologies is an extremely active area of research," says Nienhaus. "Fluorescent proteins are of particular importance in this context, as they can be excellently used to label living cells and organisms."
To do so, he and his team "fish" genes of fluorescent proteins from marine organisms, further optimize them and infiltrate them into other cells and organisms. These cells then produce the luminescent proteins – often as appendages of other proteins that can be specifically observed on the basis of light emission. Some of these proteins can be precisely activated via light irradiation at specific wavelengths. This "turning on and off" is an important step for highest-resolution imaging. "You can still also selectively make subcellular structures or individual organs glow in certain colors," Nienhaus says.
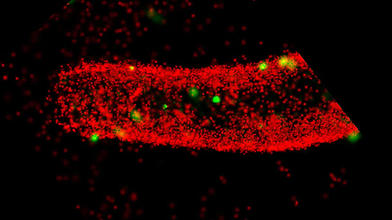
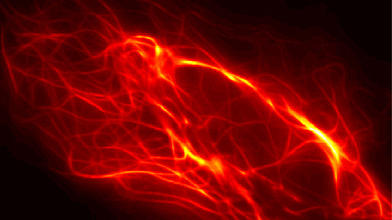
Biophysicist Nienhaus and his team are using their microscopic techniques to record movies that show the development of zebrafish embryos in 3D over many hours. This allows them to track the migration of each cell during development and, for example, analyze disease-related maldevelopments. "Our self-developed, ultra-high-resolution fluorescence microscopes and new marker technologies enable us to record images and movies that show biomolecular processes much more precisely than conventional technologies currently used in biological and biomedical research," says Professor Nienhaus: "This will open doors to completely new insights and applications in life sciences, medicine and pharmacology."